Research Article - Der Pharma Chemica ( 2021) Volume 13, Issue 3
Synthesis of Cefixime Nanoparticles an Attempt to Enhance Their Development and Validation of Spectrophotometric Methods for the Determination of Pharmaceutical Forms
Basavaraj Hiremath1*, Nagesh Gunavanthrao Yernale2,3 and B.H.M. Mruthyunjayaswamy32Department of Chemistry, Guru Nanak First Grade Science, Commerce and Post Graduate College, Bidar – 585 403, Karnataka, India
3Department of Post Graduate Studies and Research in Chemistry, Gulbarga University, Kalaburagi – 585 106, Karnataka, India
Basavaraj Hiremath, Department of Chemistry, S. S. Margol Degree College, Shahabad – 585 228, Karnataka, India, Email: drbhiremath25@gmail.com
Received: 07-Feb-2021 Accepted Date: Mar 21, 2021 ; Published: 30-Mar-2021
Abstract
Cefixime nanoparticles were prepared by anti-solvent precipitation with syringe pump (APSP) method. Two simple and sensitive spectrophotometric methods I and II for the analysis of cefixime nanoparticles in either pure form or their pharmaceutical formulations are described. Method I is based on the oxidation of the drug with ferric ion succeed by complex development reaction with 1, 10-phenanthroline (1, 10-PTL) to obtain orange-red colored chromogen shows λmax at 510 nm also the method II on the reaction between diazotized drug with diphenylamine (DPA) in neutral medium to yield a pink-colored product which has λmax at 512 nm. The products are unchanging for more than 10 and 4 hrs respectively. Common excipients used as additives in pharmaceutical dosage do not impede the proposed methods. Thus the proposed methods proved to be easy, low cost, and accurate, are highly reproducible and have been applied to a broad range of pharmaceutical preparations and the outcome compares favorably with those of the official method. Prepared nanoparticles samples showed a better outcome than bulk drug samples.
Keywords
Cefixime; Nanoparticles; 1, 10-PTL; DPA; Dizotization; Spectrophotometry.
Introduction
Cefixime is designated chemically as (Z)-7-[2-(2-Aminothiazol-4-yl)-2-(carboxymethoxyimino) acetamido]-3-cephem-4-carboxylic acidtrihydrate [1,2]. Cefexime is an antibiotic, and a 3rd generation cephalosporin is extremely steady in the presence of beta-lactamase enzymes [3]. Cefexime is used in the treatment of the subsequent infections stimulated by vulnerable strains of the selected microorganism; uncomplicated urinary tract infectivity caused by Escherichia coli and Proteus mirabilis, otitis media caused by Haemophilus influenza (beta-lactamase positive and negative strains), Moraxella catrrhails mainly of which are beta-lactamase positive, and S. pyogenes, pharyngitis and tonsillitis induced by S. pyogenes, acute bronchitis, acute exacerbations of chronic bronchitis induced by Streptococcus pneumonia and Haemophilusinfluenzae and uncomplicated gonorrhea caused by Neisseria gonorrhea [4]. Cefixime was used as a model drug [5] and it has a broad antibacterial spectrum against various Gram-positive and Gram-negative bacteria, including Haemophilusinfluenzae, Neisseria gonorrhoeae, Escherichia coli, and Klebsiellapneumoniae resistant to ampicillin, cephalexin, cefaclor, and trimethoprimsulfamethoxazole. It is used for the treatment of susceptible infections, including gonorrhea, otitis media, pharyngitis, lower respiratory-tract infections such as bronchitis, and urinary-tract infections [6-8].
Literature survey reveals that very few methods like spectrophotometric [9-11], high-performance liquid chromatography (HPLC) [12-14], high-performance thin-layer chromatography (HPTLC) [15-17], LC-MS [18,19], high-performance capillary electrophoresis [20,21] and spectrofluorimetric methods [22-24] are available for the analysis of cefexime. Various most recent drugs are of slight water solubility and little dissolution rates [25]. Their solubility and dissolution rapidity can be rising by reducing particle size [26,27]. Reducing the size of the particles will increase surface area, which can raise the speed of dissolution in aqueous resembling body fluids [28-30]. The dissolution speed is straightway proportional to the exposed surface area to the medium used in dissolution [31]. The small size of nanoparticles means that they contain disparate physic-chemical and physiological properties relate to outsized particles, such as condensed light scattering, enhanced stability to gravitational separation and aggregation, quicker dispersion rates, elevated solubility, and higher diffusion rates due to biological barriers [32].
The high surface/volume ratio increases the importance of the properties of the surface molecules over the bulk molecules [33]. There are two different strategies are used for the preparation of nanoparticles. These are top-down methodology in which larger size particles split down into nanoparticles and bottom-up approach in which smaller particles increased collectively up to nanosize [34]. In general, more energy is required in the top-down methods rather than bottom-up methods [35]. Milling and homogenization be the two conventional top-down attempts, in which unrefined particles are crushed down into undersized particles [36,37]. In comparing the bottom-up method, such as antisolvent precipitation, supercritical fluid tools and spray freezing into a liquid are seldom in use. In comparison to milling and high-pressure homogenization, a few bottom-up methods like antisolvent precipitation are quite effortless, price-effective, and trouble-free for scaling up [38,39].
The preparation of nanoparticles by the most used bottom-up method is solvent/antisolvent precipitation [40]. It is an excellent method in which there is no necessity of precise apparatus and intricate working setting the costs are comparatively low, the process might hold up and the threat of test is significantly slighter as a contrast to a top-down method. This process is moreover used in the pharmaceutical industry [41]. Nanoparticles are additionally useful for drug delivery, in particular for high hydrophobic agents, and can enhance their low water solubility and dissolution rapidity [42].
With the significant usage of lower size particles in drug dissolution and bioavailability, the current study aims to increase the dissolution speed and validation of cefixime in pure and various pharmaceutical firms by the preparation of condensed size particles (nanoparticles) with the help of an antisolvent precipitation technique. The ease of the current methods is that the reagents used in both are simply available and the chemistry of the reagents is previously well recognized. The reactions involved with these reagents are easy, quick and sensitive in the ranges of determinations compared with former established methods. Further, spectrophotometric methods involve simple instrumentation which is priced effective compared with other instrumental techniques, which ordinary laboratories cannot afford to have. The present methods involve the formation of highly colored species that are stable for 10 and 4 hrs respectively which makes it easier for their determination. The idea of the present work is to produce a simple, sensitive and rapid spectrophotometric determination of cefixime nanoparticles and the methods are free from interference when excipients are present. In continuation of our research work on drug analysis we hereby report two sensitive spectrophotometric methods for the assay of cefixime in either pure form or pharmaceutical forms. The methods are based on the oxidation of drug with ferric ion followed by complex formation reaction with 1, 10-phenanthroline and the coupling reaction between the diazotized drug and diphenylamine (DPA) in a neutral medium.
Experimental
Apparatus
An ELICO Model SL-164 Double Beam UV-visible spectrophotometer and 1.0 cm matched quartz cells were utilized for absorbance measurements.
Reagents and Materials
Drug (Gift sample from Alkem laboratories Ltd, Daman), 1, 10-PTL (Merck), DPA (S.D.Fine), NaNO2 (A.R), FeCl3 (A.R), o-phosphoric acid (A.R) and hydrochloric acid (A.R) were used. All other chemicals and solvents used were of analytical reagent grade. Deionized water was used to prepare all solutions and in all experiments. Commercial dosage forms were purchased from local sources.
Solutions
Stock solutions were prepared by accurately weighing 100 mg of cefixime into a 100 ml calibrated flask, dissolved in 100 ml deionized water and kept in dark to avoid any degradation of the drug. The working standard solution of cefixime containing 100 μg/ml was prepared by further dilution. A 0.03 M alcoholic solution of 1, 10-phenanthroline and 0.25 % alcoholic solution of diphenylamine were freshly prepared. About 0.03 M FeCl3 solution, 0.01 M HCl, 0.1 % aqueous solution of NaNO2 and 0.2 % aqueous solution of ammonium sulfamate were used.
Procedure
In method I, aliquots of the working standard solution (0.2-1.0 ml) of cefixime (1.0 ml=100 μg) were transferred into 10 ml volumetric flasks. To each, 0.2 ml of 0.03 M FeCl3 and 0.5 ml of 0.03 M 1, 10-phenanthroline solution were added, heated on the water bath for 10 min and then cooled to ambient temperature. To each one flask 2.0 ml of 0.02 M o-phosphoric acid solution was added then the final volume was brought up to 10 ml with deionized water. The solutions were mixed completely and the absorbance was studied at 510 nm against reagent blank and a calibration graph was constructed.
In method II, aliquots of the working standard solution (1.0-5.0 ml) of cefixime (1.0 ml=100 μg) were added into a succession of 10 ml volumetric flasks. To each one of the above flask, 1.0 ml of 0.01 M hydrochloric acid and 0.5 ml of 0.1 % NaNO2 were added and put to one side for 10 min at room temperature. An aqueous solution of 0.5 ml of 0.2 % ammonium sulfamate and an alcoholic solution of 1.0 ml of 0.25 % diphenylamine was then added to each flask and diluted to 10 ml with deionized water. The absorbance of the pink-colored species was measured at 512 nm against reagent blank and the amount of cefixime was computed from its calibration graph [43].
Preparation of cefixime nanoparticles
Anti-solvent precipitation with syringe pump (APSP) method was employed for the preparation of cefixime nanoparticles [44]. In this method, a saturated solution of cefixime was prepared in 50 ml of methanol. The syringe was made full with the standard drug solution which was instantly introduced at a specific flow rate (2 ml/min) into antisolvent (deionized water) of definite volume with stirring rate (9,000 rpm), the ratio of 1:15 volumes of saturated drug solution and deionized water. After stirring the resulting mixture was evaporated quickly using a rotary evaporator to get nanoparticles of the drug which were used as such for further analysis.
Procedure for the analysis of the cefixime nanoparticles in commercial samples
Tablets:
Ten to twenty tablets depending on content per tablet were weighed and mixed thoroughly. Preparation of cefixime nanoparticles from pharmaceutical forms was obtained by Anti-solvent Precipitation with Syringe Pump (APSP) method. An amount of the powder corresponding to 100 mg of the active constituent was weighed and transferred into a 100 ml volumetric flask. About 60 ml of deionized water added and shaken thoroughly for nearly 20 min and the volume was increased to the mark with deionized water shaken, filtered using quantitative filter paper. For spectrophotometric determination, the filtrate was diluted consecutively to obtain 100 μg/ml for the drug. A suitable portion was then used for analysis.
Results and Discussions
In method I, the drug was diazotized in an acidic medium and coupled with DPA in a neutral aqueous medium. In method II, the drug was oxidized by Fe3+ ion followed by a complex formation reaction with 1, 10-phenanthroline to form a ferrion chelate.
Spectral characteristics
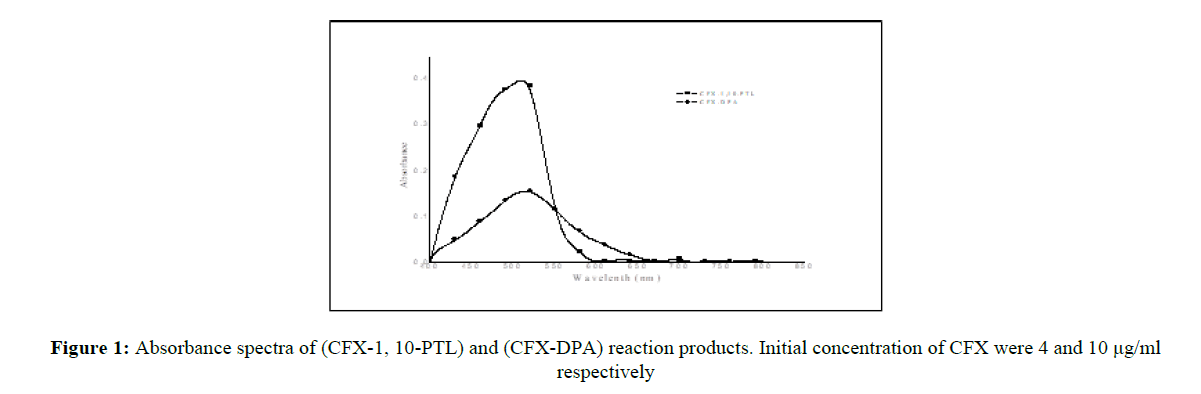
The absorbance spectra of (CFX-10-PTL) orange-red-colored product with λ max at 510 nm and of the pink-colored product (CFX-DPA) with λ max at 512 nm are shown in (Figure 1). The above-mentioned reagent blanks have practically negligible absorption in both systems.
Optimum reagent concentration
For the method I, 0.03 M FeCl3 in the range of 0.2-1.0 ml, 0.2-1.0 ml of 0.03 M 1, 10-phenanthroline and 1.0-5.0 ml of 0.02 M o-phosphoric acid solution were necessary for the development of maximum color intensity.
For the method II, 0.01 M HCl in the range of 0.5-2.5 ml, 0.5-2.5 ml of 0.1 % NaNO2, 0.5-2.5 ml of ammonium sulfamate solution and 0.2-1.0 ml of 0.25 % DPA solution were necessary for the development of maximum color intensity.
Hence, required volumes of all the reagent solutions were used as mentioned in the recommended procedure. The surplus of nitrite could be reduced by the addition of 0.5 ml of 0.2 % ammonium sulfamate solution. A surplus of ammonium sulfamate has no cause on the colored solution with a different solvent like water, methyl alcohol, ethyl alcohol, glacial acetic acid and acetonitrile which have been tested. However, dilution up to the mark with water imparted the highest intensity and steadiness of the color.
Quantification
Beer’s law was obeyed on the CFX concentration limit of 2-10 μg/ml, 10-50 μg/mlfor the method I and II respectively. The proposed procedures are validated by determining various optical parameters which are listed in Table 1. The linearity, slope and the intercepts have been calculated using the regression equation y = ax + b, where ‘y’ represents optical density, ‘x’ the concentration of drug in μg/ml and ‘a’ and ‘b’ represent slope and intercept, respectively. Precision and accuracy of the intended methods were tested by carrying out the determination of eight replicates of pure and pharmaceutical dosage of the drug, whose concentration lies within Beer’s law range. The values of standard deviation (S.D.), relative standard deviation (R.S.D.) and range of error at 95 % confidence level were calculated.
| Parameter / characteristics | Method I | Method II |
|---|---|---|
| Color | Orange-red | Orange-red |
| λ max (nm) | 510 | 512 |
| Stability (hrs) | 10 | 4 |
| Beer's law range (μg/ml) (C) | 10-Feb | Oct-50 |
| Limit of detection (μg/ml) | 0.091 | 0.68 |
| Limit of quantification (μg/ml ) | 0.3 | 2.25 |
| Molar absorptivity (l mol-1/cm) | 3.584×104 | 3.97×103 |
| Sandell's sensitivity (μg/cm2) | 0.01 | 0.45 |
| Regression equation(Y)a | ||
| Slope (a) | 2.4761×10-3 | 6.0262×10-2 |
| Intercept(b) | 9.9869×10-2 | 9.0881×10-3 |
| Correlation co-efficient (r) | 0.9999 | 0.9996 |
| R.S.D. (%)b | 0.2952 | 0.7041 |
| % Range of error’s b | ± 0.0014 | ± 0.0019 |
a Y = ax + b, where ‘x’ is the concentration in μg/ml. b Eight replicates
Table 1: Parameter for the spectrophotometric estimation of drug
There was no altering in λ max when the least concentration of the analyte was estimated, for the calculation of LOD,LOQ was found to be three times that of LOD which is in concurrence with Thumb’s rule. The experiment for the planned methods was carried out by the 2ndanalyte on dissimilar days and the results produced justified the ruggedness of the proposed methods. The two methods were applied to many pharmaceutical formulations and a recovery study was constructed. The optical characteristics and precision data for the methods (I and II) recommended are available in (Table 1).
Reaction sequence
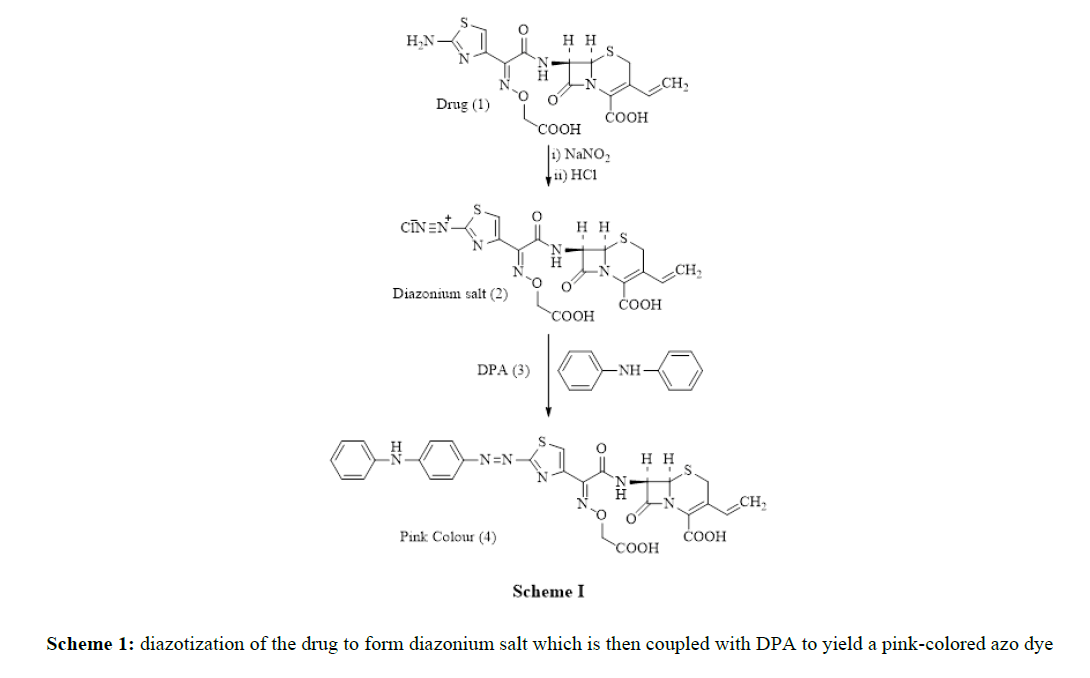
Method I, describes the oxidation of the drug with Fe3+ ion pursued by complex formation reaction of Fe2+ ions with 1, 10-phenanthroline to obtain the orange-red-colored product. Method II, describes the diazotization of the drug to form diazonium salt which is then coupled with DPA to yield a pink-colored azo dye. The reaction mechanism of method II is shown in Scheme 1.
Stability
The colored species obtained in the proposed methods (I and II) were stable for 10 and 4 hrs respectively at room temperature. In method I the reaction of colored complex formation was gradual at room temperature (25ºC) and required more time for reaction completion. Hence, efforts were made to quicken by carrying out the reaction at elevated temperatures. It was noticed that the maximum color intensity was attained by heating the reaction mixture at 80 ºC on a water bath for 10 min. The absorbance’s remained constant at room temperature for more than 8 hrs. In method II the color was stable for more than 4 hrs at room temperature.
Interference
A comprehensive study on the interference of different associated substances on the determination of the drugs was made. For method I, 4 μg/ml CFX was selected to check the interference. For method II, 10 μg/ml CFX was used to analyze the interference. Before adding the reagents, a known quantity of the interfering material was added and the reaction was conducted for the methods (I and II). The level of interference by various excipients that repetitively accompany the pharmaceutical formulations are tabulated in (Table 2). Amines such as aniline, piperidine, morpholine, etc. interfere in the DPA method, because of the diazotization reaction. The extent of interference by excipients that often accompany the pharmaceutical preparations have been studied for both methods. It was found that methods (I and II) achieved incomparable results for the determination of pure CFX in presence of excipients that do not interfere in the methods (I and II). The results are given in (Table 2). The error of 2.0 % in the absorbance reading was considered tolerable.
| Material | Amount (mg) | % Recovery of drug ± R .S. D a. | |
|---|---|---|---|
| Method I | Method II | ||
| Magnesium stearate | 30 | 99.80 ± 0.13 | 99.69 ± 0.40 |
| Lactose | 30 | 99.73 ± 0.16 | 99.64 ± 0.16 |
| Dextrose | 30 | 99.57 ± 0.12 | 99.64 ± 0.26 |
| Starch | 30 | 99.81 ± 0.13 | 99.56 ± 0.19 |
| Gum acacia | 30 | 99.66 ± 0.49 | 99.45 ± 0.19 |
| Talc | 30 | 99.56 ± 0.29 | 99.52 ± 0.37 |
| Carboxy methyl cellulose | 30 | 99.74 ± 0.19 | 99.70 ± 0.40 |
| Sodium alginate | 30 | 99.66 ± 0.31 | 99.58 ± 0.23 |
Method I, * 4 μg/ml of drug taken.Method II, * 10 μg/ml of drug taken.aAverage of five determinations.
Table 2: Determination of cefixime in the presence of excipient and other substances.
Applications
The uses of the current methods were examined for cefixime nanoparticles pharmaceutical preparations. The results of the analysis of drugs in pharmaceutical dosage forms are given in (Table 3). The similar lot tablets were also analyzed by the official method [45]. A statistical analysis of the results using Student t-test and F-test values were compared with the official method. Mean values were obtained in the Student t- and F- test at 95 % confidence limits for seven degrees of freedom [46].
| Preparationsa | Label claim mg/Tablet | Foundb (recovery ± S D) | Students t-valuec | F-valued | ||||
|---|---|---|---|---|---|---|---|---|
| Method I | Method II | Reference method | I | II | I | II | ||
| Taxim-O (1) | 50 | 99.8 | 99.52 | 99.85 | 0.21 | 1.28 | 1.46 | 1.04 |
| ± 0.43 | ± 0.51 | ± 0.52 | ||||||
| Mahacef | 100 | 99.52 | 99.78 | 99.92 | 2.07 | 0.71 | 1.05 | 1.05 |
| ± 0.38 | ± 0.40 | ± 0.39 | ||||||
| Ominicef-O (3) | 200 | 99.74 | 99.76 | 99.79 | 0.18 | 0.11 | 2.95 | 2.54 |
| ± 0.39 | ± 0.42 | ± 0.67 | ||||||
a. Marketed by: 1, Alkem; 2, Mankind and 3, Aristo. b. Mean value of eight determinations c. The tabulated value at 95% confidence level is 2.365 d. The tabulated value at 95% confidence level is 3.79.
Table 3: Results of an assay of cefixime nanoparticles in pharmaceutical formulations.
The results reveal no significant variation concerning to accuracy and precision. Tablets containing 50, 100 and 200 mg of drug samples labeled as 1, 2 and 3 respectively, were analyzed by the current methods and the accuracy was assessed by the standard addition method in which variable amounts of the pure drug was added to the previously analyzed portion of cefexime nanoparticles of pharmaceutical formulations. Results are recorded in (Table 4) and confirmed that the current methods are not liable to interference by fillers usually formulated with standard drugs. The synthesized nanoparticles were found to show sensitive, rapid, simple and precise than the bulk drug.
| Method I | Method II | |||||||
|---|---|---|---|---|---|---|---|---|
| Formulationsa | Amount of CFX nanoparticles in formulation/mg | Amount of drug added/ | Total found/mg | % Recovery of pure drug b | Amount of CFX nanoparticles in formulation/mg | Amount of drug added/ | Total found/mg | % Recovery |
| mg | mg | of pure | ||||||
| drug b | ||||||||
| 1 | 5.94 | 3 | 8.92 | 99.33 | 2.96 | 5 | 7.94 | 99.6 |
| 5.94 | 6 | 11.91 | 99.5 | 2.96 | 10 | 12.95 | 99.9 | |
| 5.94 | 9 | 14.93 | 99.88 | 2.96 | 15 | 17.95 | 99.93 | |
| 2 | 3.98 | 3 | 6.99 | 100.33 | 3.01 | 5 | 8 | 99.8 |
| 3.98 | 6 | 9.96 | 99.66 | 3.01 | 10 | 13.01 | 100 | |
| 3.98 | 9 | 12.97 | 99.88 | 3.01 | 15 | 17.99 | 99.86 | |
| 3 | 4.02 | 3 | 7.01 | 99.66 | 5.98 | 5 | 10.96 | 99.6 |
| 4.02 | 6 | 9.99 | 99.5 | 5.98 | 10 | 15.99 | 100.1 | |
| 4.02 | 9 | 13.03 | 100.11 | 5.98 | 15 | 20.97 | 99.93 | |
a.Branded by: 1, Taxim-O (50 mg) 2, Mahacef (100 mg) and 3, Ominicef-O (200 mg). b. Average of three determinations
Table 4: Results of recovery studies by the standard-addition technique
Conclusions
The enhancement in sensitivity of assay and recovery study is due to the drug nanoparticles and exhibited better results than the bulk drug. The current methods for the estimation of CFX nanoparticles in pharmaceutical forms are simple, rapid and sensitive. The methods do not require heating or extraction. The statistical data and the recovery study data specify the reproducibility and accuracy of the methods (I and II). The methods could be used for the determination of CFX in quality control laboratories.
Acknowledgment
The authors are sincerely very thankful to The Chairman, Department of Post Graduate Studies and Research in Chemistry, Gulbarga University, Kalaburagi, for providing laboratory facilities.
References
- Sweetman SC, Pharmaceutical press. 2002, p.240.
- O’ Niel, MJ, Ed, Merck Index Merck and Co. Inc. Rathway, 2001, p.160.
- Nath AP, Balasubramanian A and Ramalingam K. Int J Res Pharm Sci. 2020, p.623.
- Bluestone and Charles D. Md., Pediatr. Infect. Dis. J., 12: 1993.p.75.
- Tan BJ. Can J Infect Dis. 1995, 6: p.204.
- Brogden RN and Richards DM. Drugs. 1989, 38: p. 524-550.
- Sweetman SC. Pharmaceutical Press, 2002.
- Suganthi M and Kuppanagounder PE. Phys Chem Liq. 2018, p. 299-310,
- Mali AD, Int. J. Pharm. Pharm. Sci, 2015, 7: p. 321-325.
- Ramadan AA, Mandil H and Dahhan M., Int J Pharm Sci. 2013, 5: p. 428-433.
- Ramadan AA, Mandil H and Dahhan M. Asian J Chem. 2013, 25: p. 3457-346.
- Khandagle KS, Gandhi SV, Deshpande PB et al., Int J Pharm Sci. 2011, 3: p. 46-48.
- Arshad HM, Gauhar S, Bano R et al., J Pharm Sci. 2010, 2: p. 53-65.
- Khan A, Iqbal Z, Khan MI et al., J Chromatogr B. 2011, 879:p. 2423-2429.
- Pawar SJ, Kale AP and Amrutkar MP et al., Asian J Res Chem. 2010, 3: p. 299-301.
- Rao J, Sethy K and Yadav S., Int J Compr Pharm. 2011, 2: p. 1-4.
- Meng F, Chen X, Zeng Y et al., J Chromatogr B. 2005, 819: p. 277-282.
- Talebpour Z, Pourabdollahi H, Rafati H et al., Sci Pharm. 2013, 81: p. 493-504.
- Honda S, Taga A, Kakehi Ket al., J Chromatogr A. 1992, 590: p. 364-368.
- Alnajjar AO, J. Liq Chromatogr RT. 2013, 36: p. 2687-2697.
- Shah J,Jan MR and Shah S, J Fluoresc. 2011, 21: p. 579-585.
- Manzoori JL,Amjadi MN, Soltani A et al., Iran J Basic Med Sci. 2014,17: p. 256-262.
- Abdollahi AA and Bavili-Tabrizi, Pharm Sci. 2015, 22: p. 28-34.
- Khandagle KS, Gandhi SV, Deshpande PB et al., Int J Pharm Sci. 2011, 3: p. 46-48.
- Aljerf L and AlMasri N., Int J Drug Disco. 2018, 2: p. 10.
- Abhilash M, Int J Pharma Bio-Science. 2010, 1(1): p. 53.
- Thorat Y, Gonaraj D and Hosamani A., Int J Phram Sci Res. 2011, 2(10): p. 2501.
- Wang JX and Zhang QX, Chem Eng J. 2010, 162(2): p. 844.
- Overhoff KA, Jhonson RO and William RO, Polymeric Drug Delivery II. 2006, p.305.
- Liversidge GG and Cundy CK. Int J Pharm. 1995, 125: p. 91.
- Noyes AA and WhiteneyWR., J Amer Chem Society. 1997, 19(12): p. 930.
- Aljerf L and AlHamwi B., Madridge. J Nanotechnol Nanosci. 2018, 3: p. 118.
- Christain P, Kammer FV and Baalousha M, Th. Hofmann, Ecotoxicology, 2008, 17(5): p. 326.
- Biswas A, Bayer IS, Biris AS et al., Adv Colloid Interface Sci. 2012, 170: p. 2.
- Kesisogolu F, Panmai, S. and Wu, Y., Adv Drug Del Rev. 2007, 59(7): p. 631.
- Panagiotou T and Fisher RJ, Functional Food in Health and Disease. 2013, 3: p. 274.
- Hixon L, Prior M and Prem H., Chemical Eng J. 1990, 97: p. 94
- Horn D and Rieger J, Angewandate Chemie-Int Edn. 2001, 40(23): p. 4330.
- Rogers TL and Gillespie IB, Hitt JE, Phram Research. 2004, 21(11): p. 2048.
- de Waard H, Frijlink HW and Hinrichs WL. Pharm Res. 2011, 28: p. 1220.
- Thorat AA and Dalvi SV. Chem Eng J. 2012,181: p. 1.
- ReverchonE, The J. Supercritical Fluids, 1999, 15(1): p. 1.
- Shankar DG, Sushma K, Laxmi RV et al., Indian Drugs. 2001, 38: p. 617.
- Kakaran MN, Sahoo G, Tan IL et al., J Nanoparticles Res. 2012, 14(3):p. 757.
- Indian Pharmacopeia, Controller of Publications, Delhi. 1996, 1: p.149.
- Miller JC. Statistic in Analytical Chemistry, Ellis Horwood, Chichester, 13th Edn., 1993.